Arsenic is a naturally occurring element present in ground waters in many parts of the world. Natural mineral deposits, agriculture and industrial pollution all are factors that contribute to the presence of arsenic in water. It has been well established that arsenic is toxic at high concentrations; however, recent research has shown that relatively low concentrations can cause adverse health effects, including cardiovascular problems, diabetes and various forms of cancer. For more than 50 years, the EPA-mandated maximum contaminant level (MCL) for arsenic in drinking water has been 50 parts per billion (ppb). However, as of January 2006, the acceptable level will be reduced to 10 ppb; it has been estimated more than 5 percent of all U.S. water supplies will be out of compliance.
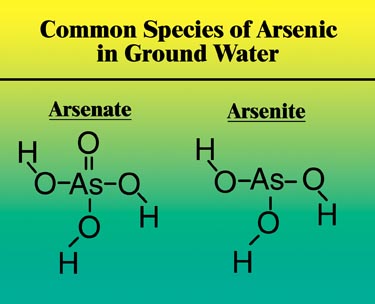
Arsenic is stable in several oxidation states, but the two forms most commonly found in water are arsenate (As5+) and arsenite (As3+)(see Figure 1). Arsenic occurs in the arsenate form in most surface waters and oxidizing conditions, while in anaerobic ground waters and reducing conditions, arsenic exists mainly as arsenite. Arsenite can be oxidized to arsenate with oxidizing agents such as oxygen, chlorine or potassium permanganate. Since arsenate is more easily removed than arsenite, oxidation before treatment is the preferred protocol for waters containing high levels of arsenite.
Treatment Processes
Several processes have been developed for the removal of arsenic from drinking water. A brief overview of the most common technologies is given below:
Coagulation/Filtration
A chemical reagent (such as lime, ferric hydroxide or alum) that forms insoluble arsenic compounds is added to the contaminated water; these arsenic compounds then can be removed by sand filtration. This method of arsenic removal originally was used for treating mining effluents where the arsenic is present at high concentrations, however it has been shown effective with drinking water in certain instances as well.Three problems with using such treatment of drinking water are:
(1) the treatment chemical often affects the pH of the treated water (this is especially true for the most effective chemical - ferric chloride);
(2) the dosage of coagulant chemical has to be fairly high (3 mg/l to 5 mg/l ferric chloride for 50 ppb arsenic); and
(3) a relatively large volume of waste sludge is produced.
Ion Exchange on Polymeric Resins
Ion exchange resins work simply by exchanging one ion from the resin surface for a like-charged ion in the water passing over it. This technique can work well in some instances, but usually is poor in its selectivity. For anion exchange resins, sulfate, selenium and nitrate all are preferred over arsenic, and their presence therefore will reduce the resin's arsenic adsorption capacity. Additionally, the effectiveness of ion exchange often is severely affected by the presence of high salt concentrations. Ground and surface waters frequently have high levels of inorganics, which makes this method unsuitable as an effective method for treating such waters. Disposal of spent brine regenerant solution also may be problematic.Reverse Osmosis/ Nanofiltration
This method is extremely effective in removing (“rejecting”) salts and ions (including arsenate), but it is not selective.
The rejection of scale-causing ions such as calcium leads to the precipitation of these materials on reverse osmosis (RO) and nanofiltration (NF) membranes. This typically results in fouling of the membrane surface and must be avoided by adding anti-scaling chemicals and/or acid washing. Additionally, RO and NF processes often have a poor recovery rate (i.e., often 50% or more of the incoming water is wasted). Another disadvantage is that large amounts of concentrate waters are generated and must be disposed.
Adsorption
Adsorption of arsenic normally is achieved by filtering arsenic-contaminated water through a column of granular arsenic-adsorbing media.For decades, activated alumina (Al2O3) was the media of choice for adsorption of arsenic. However, this media's performance is very pH-dependent (pH of 5.5 is required for best performance) and is affected by high concentrations of fluoride, sulfates and chlorides. The possibility of aluminum leaching into water also may be a concern.
Granular ferric hydroxide and granular ferric oxide are two types of adsorptive media developed by German companies to take advantage of arsenic-ferric ionic attraction. Advantages of these media include a relatively high operating pH range (up to 8) and good adsorption capacity. However, they cannot be regenerated (single-use) and are negatively affected by the presence high concentrations of iron and silica. There also is arsenic-removal media available on the market that is unaffected by fluorides, chlorides, sulfates and iron, and can be regenerated for re-use after arsenic saturation.
In summary, there are several options available for arsenic treatment, and no single technology is best for every application. Water chemistry, speciation of arsenic (arsenate or arsenite), daily water usage, and maximum flow rate all must be considered before selecting a technology. Equally important are the qualifications of the technology provider: Is the technology patented? Is it NSF-certified? Does it have third-party (e.g., ETV) performance verification? What sort of track record does the company have and how long has it been in business? With a little background knowledge and by asking the right questions, you can improve your chances of getting the technology that's right for you and your customers.
ND
Report Abusive Comment